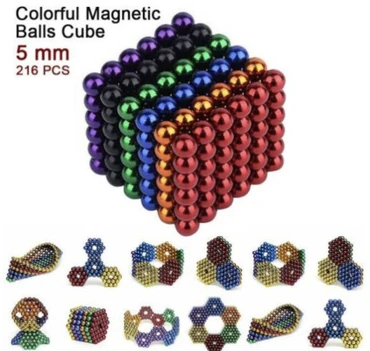
Joybuy of Irvine, Calif., is recalling about 4,240 Relax 5mm Science Kit, Large Hematite Magnets Magnetic Stones Building Blocks.
The magnetic ball sets do not comply with the requirements of the mandatory federal magnet regulation as they contain one or more magnets that fit within the Consumer Product Safety Commission’s small parts cylinder, and the magnets are stronger than permitted.
When high-powered magnets are swallowed, they can attract each other, or another metal object, and become lodged in the digestive system. This can result in perforations, twisting and/or blockage of the intestines, infection, blood poisoning, and death.
CPSC estimates 2,400 magnet ingestions were treated in hospital emergency departments from 2017 through 2021. The agency is aware of seven deaths involving the ingestion of hazardous magnets, including two outside the U.S.
This recall involves 5mm magnetic balls which are small, spherical, loose and separable neodymium rare-earth magnets with a strong magnetic flux.
The magnets are sold in a set of 216 multi-colored magnetic balls encased in a clear, plastic case and a portable, tin storage box. The set includes a black storage bag.
The magnetic ball sets, manufactured in China, were sold exclusively online at Walmart.com from February 2022, through April 2023, for between $14 and $15.
What to do
Consumers should stop using the recalled magnetic balls immediately, take them away from children and contact Joybuy to receive a pre-paid label to return them for a full refund.
Joybuy is notifying all known purchasers directly.
Consumers may contact Joybuy Marketplace Express collect at (302) 426-4543 from 9 a.m. to 5 p.m. (PT) Monday through Friday, by email at oversea-service@jd.com, or online at https://www.walmart.com/seller/16214 for more information.
Joybuy of Irvine, Calif., is recalling about 4,240 Relax 5mm Science Kit, Large Hematite Magnets Magnetic Stones Building Blocks.
The magnetic ball sets do not comply with the requirements of the mandatory federal magnet regulation as they contain one or more magnets that fit within the Consumer Product Safety Commission’s small parts cylinder, and the magnets are stronger than permitted.
When high-powered magnets are swallowed, they can attract each other, or another met...